Leave Your Message
Request a Quote
Protein purification is essential in biochemistry and biotechnology. It enables researchers to isolate specific proteins for study or application. Dr. Jane Smith, a renowned expert in this field, notes, “Effective protein purification requires finesse and precision.” Her words highlight the challenges that come with this intricate process.
The techniques in protein purification are varied. Methods such as chromatography or precipitation are often employed. Each technique has its own benefits and drawbacks. The choice of method depends on the protein's characteristics and the desired purity level. Yet, achieving optimal results can be frustrating. Sometimes, impurities linger despite best efforts.
Trial and error is common in protein purification projects. Many factors can affect the outcome. Temperature, pH, and salt concentrations often need adjustments. Researchers must remain vigilant and flexible. Embracing these challenges is part of mastering protein purification. Ultimately, the pursuit of pure proteins drives innovation in science and industry.

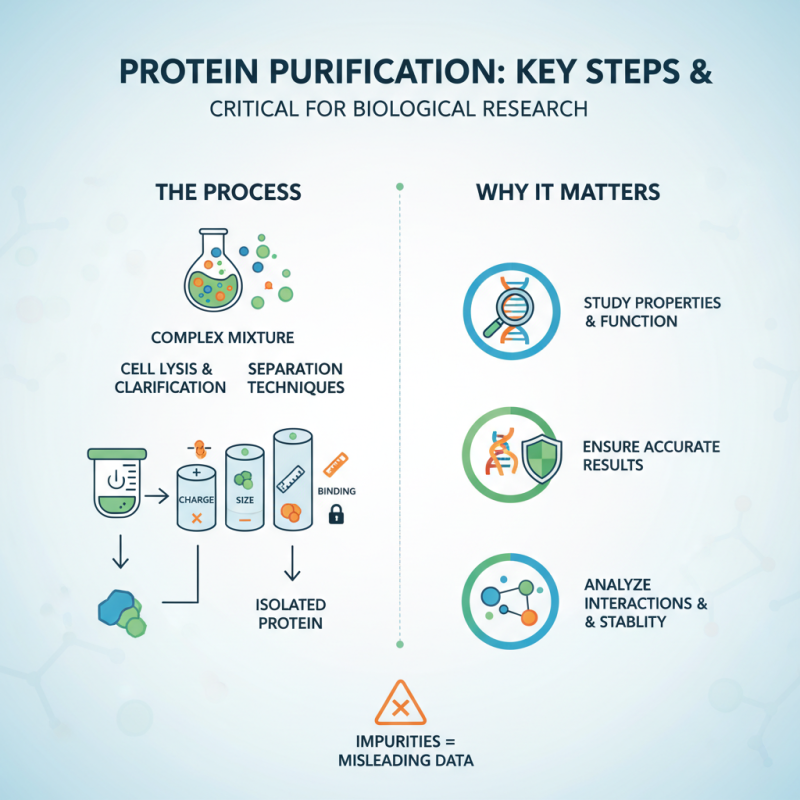
Protein purification is a critical step in biological research. It allows scientists to isolate specific proteins from complex mixtures. This process helps in studying properties and functions of proteins in detail. Without proper purification, results can be misleading. Impurities can affect activity, stability, and interactions.
Challenges often arise during purification. Techniques may not yield pure samples. Some proteins are difficult to extract due to their properties. For instance, membrane proteins can be tricky to work with. Scientists must adapt methods based on protein nature and source. Experimentation is key. Each step in purification carries the risk of losing target proteins, making it vital to optimize every condition.
Effective protein purification fosters advancements in various fields. It supports drug development and disease understanding. When researchers invest time in perfecting purification techniques, the benefits can be significant. However, achieving the ideal purification remains a work in progress, reminding us that science is continually evolving.
Protein purification is a critical step in many biological studies. However, several challenges can hinder this process. One significant issue is the low yield of purified proteins. Often, proteins are lost during the purification steps. This can lead to insufficient material for subsequent experiments. Researchers need to optimize their protocols to minimize losses. Each step must be carefully assessed.
Another common challenge is achieving the desired purity. Contaminants can co-purify with the target protein. This can affect the results of downstream applications. Specific techniques, such as affinity chromatography or ion exchange, may not be suitable for all proteins. Finding the right method requires experimentation and adjustments. Sometimes, the process can seem tedious and frustrating.
Finally, protein stability is a major concern. Many proteins are sensitive to changes in temperature or pH. This can complicate the purification process. Researchers often face difficulties in maintaining the stability of their proteins. Each condition must be monitored closely to avoid degradation. It’s crucial to continuously refine purification techniques to enhance efficiency and effectiveness.

Protein purification is critical in biochemistry. Various techniques exist, including affinity chromatography, gel filtration, and ultrafiltration. Each method varies in specificity, throughput, and cost. According to a recent industry report, the global protein purification market is expected to reach $5.2 billion by 2026, driven by rising biomedical applications.
Affinity chromatography is popular for its specificity. This method uses a resin with a ligand to selectively bind proteins. However, it may not always yield pure samples. Gel filtration offers another approach, separating proteins by size. While effective, small proteins can elude detection, leading to incomplete results. Ultrafiltration serves as a great concentration technique. Yet, it can result in protein denaturation if not monitored carefully.
A study published in the Journal of Proteome Research indicates a 30% variance in purity levels across different techniques. Oftentimes, optimizing techniques requires experimentation and a bit of trial and error. Every lab might face unique challenges. Effective protein purification is not always straightforward. Understanding these methods requires diligence, illustration, and adaptability.
Effective protein purification is essential for many biological studies. There are several basic techniques that can be followed step-by-step. Here are some protocols to achieve this.
Start with suitable extraction methods. Use buffers that maintain protein stability. For instance, a phosphate-buffered saline solution often works well. Next, centrifuge your sample to separate cellular debris. The supernatant contains your protein of interest. However, clarity is vital. If the solution is murky, repeat centrifugation.
Column chromatography is another effective method. Choose a resin based on your protein's properties. Size-exclusion chromatography can separate proteins by size. Alternatively, affinity chromatography targets specific binding sites. Keep an eye on the flow rates to avoid losing samples. Patience is key, and it’s easy to rush, resulting in poor purification.
Monitor the purification process regularly. Analyze samples using techniques like SDS-PAGE. This can help assess protein purity and integrity. Inadequate purification often leads to unexpected results. It’s crucial to reflect on your methods and learn from any setbacks. Protein purification can be an art that requires practice and adjustment to perfect.
Achieving effective protein purification requires careful evaluation of both purity and yield. A well-defined quality assessment is essential to ensure that your results meet the desired criteria. One of the most common methods is SDS-PAGE, a technique that provides detailed insight into protein size and purity. By comparing the bands of your purified protein to a standard, you can immediately assess purity levels.
To enhance your evaluation process, consider performing multiple assessments. For example, spectrophotometry can also help determine protein concentration. This technique assists in quantifying yield and compares it to your original sample. Understanding the relationship between purity and yield is crucial, as high yield does not always equate to high purity.
Tip: Always document your methods and findings. This enables you to track progress and identify inconsistencies. Repeating experiments or refining techniques can often lead to better results. Maintaining a clean workspace and using appropriate reagents can also impact purity, as contaminants can easily skew your results. Always question your outcomes; are they reproducible and meaningful?







